Debunking Myths About Products for Africa: Zyomyx develops test for HIV/AIDS treatment with service-free accessory
Zyomyx Inc., a Fremont, Calif.-based company, is set to launch an innovative point-of-care CD4 test for HIV/AIDS treatment in sub-Saharan Africa in the coming months.
CD4 tests measure immune cell levels and help to determine when patients need to start or modify antiretroviral therapy. They are a crucial diagnostic component in HIV/AIDS treatment. But currently, there are significant barriers to testing for people in low- and/or middle-income countries because traditional bench-top testing equipment is only available in centralized locations and requires long wait times for results. In this system there is high loss to follow-up and many patients never receive the lifesaving antiretroviral therapy they need.
(Full disclosure: I’m the global alliance manager at Zyomyx.)
Zyomyx’s path to this point is unusual and surprising in many respects, yet its 14-year history as a Silicon Valley biotech is the underpinning of its current success designing a diagnostic tool for Africa.
In 1999 Zyomyx, with an eye on U.S. and European markets, began working on protein chip technology and pioneered the move to studying multiple proteins in parallel. The company has evolved since then, but its core competency remains the same – making complex diagnostics easy to use. President and Founding Partner Peter Wagner explains, “Even what we do today and what we are planning to do tomorrow reflects that original vision.”
Despite the fast pace of technological advances, disruptive technology takes time – patience is a critical component of the strategic vision, as Zyomyx’s leadership has discovered over the years. “Because you have to fight hard to make your product sustainable you don’t commit to product concepts that are not really vetted and tested for the demanding requirements in Africa,” said Wagner in discussing the perspective of a for-profit company operating in a space largely dominated by NGOs.
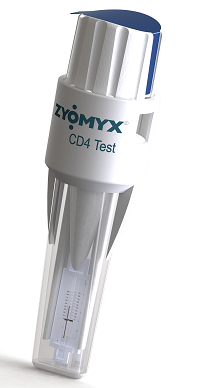
(Pictured left: Zyomyx’s CD4 test).
The process of designing the Zyomyx point-of-care CD4 product began with an early effort to understand the diagnostics market in Africa and the unmet needs. This meant debunking myths about the required specifications. As Wagner explains, “Most people think that designing products for Africa is equivalent to making them inexpensive and heat-tolerant.”
The Zyomyx team discovered early on that this view was limited. In fact, the team learned that understanding work flow, i.e., how the product is being used and the training required, is also critical. In the context of Africa, this means that instruments that require servicing and maintenance will quickly become broken pieces of expensive equipment that accumulate dust. With this knowledge, Zyomyx had to conceive a diagnostic test that would not require servicing.
Engineers and scientists came up with a self-contained test unit paired with a reusable, completely service-free accessory. In other words, the technology is all contained in the test consumable and the accessory to run the test is relatively low-tech, durable and inexpensive – if it breaks it will be disposed of and simply replaced at no cost.
The test will also deliver a result in less than 10 minutes and will cost less than (U.S.) $10 per test. Contrast this with the current standard of large, bench-top flow cytometers that require high testing volumes to reach cost efficiency, making it impossible to place instruments at community level sites with lower volumes. Flow cytometers also require batch processing, so patients must wait hours and sometimes days or even weeks for the results.
Zyomyx’s novel technology has resulted in funding from the Bill & Melinda Gates Foundation as well as a product design award from Frost & Sullivan. The company has plans to continue making complex diagnostics affordable and easy to use for a variety of health areas including infectious diseases, cancer and women’s health.
Karen Schlein is the global alliance manager at Zyomyx Inc.
- Categories
- Health Care
